Chantel Tester, Derk Joester
______________________________________________________________
Overview
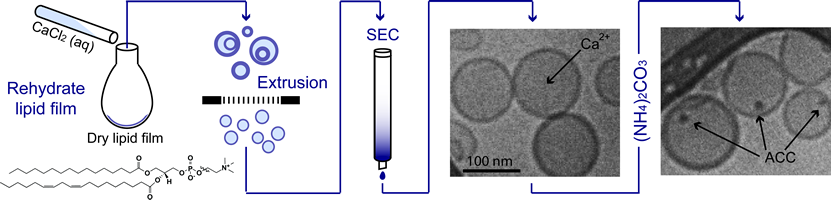
The use of metastable amorphous minerals as precursors is a widespread strategy in biomineralization that is thought to play a major role in the biological ability to control polymorph and crystal shape, introduce smooth, curving surfaces, and impart outstanding mechanical properties. This level of control is still beyond our synthetic capabilities. Unlike most in vitro syntheses performed in bulk solution, biological processing of amorphous minerals frequently occurs in "privileged environments". This compartmentalization occurs at a length scale of tens of nm to microns, and usually involves semi-permeable phospholipid bilayer membranes. Due to the small volume and large relative surface area in phospholipid vesicles (liposomes), we hypothesized that amorphous calcium carbonate (ACC) could be synthesized and stabilized by confinement and/or interaction with the membrane.

In this system, aqueous calcium salts are encapsulated within liposomes and ACC precipitation is initiated by diffusion of ammonium carbonate. Initial work has utilized simultaneous small- and wide angle X-ray scattering, X-ray absorption spectroscopy, and cryo-electron microscopy to probe the nucleation, growth kinetics, and ordering of the resulting mineral phase. We find that liposome-encapsulated ACC nanoparticles are stable against aggregation and do not crystallize for at least 20 hours. In contrast to bulk precipitation, isolated precipitation inside liposomes offers a unique platform with which to study the effects of particle size, membrane chemistry, and co-encapsulated additives on the stability of ACC. An understanding of how organisms use these parameters to control the amorphous to crystalline transition will enable enhanced crystal growth control, approaching that observed in biology.
Home | Contact Us | Sites A-Z









